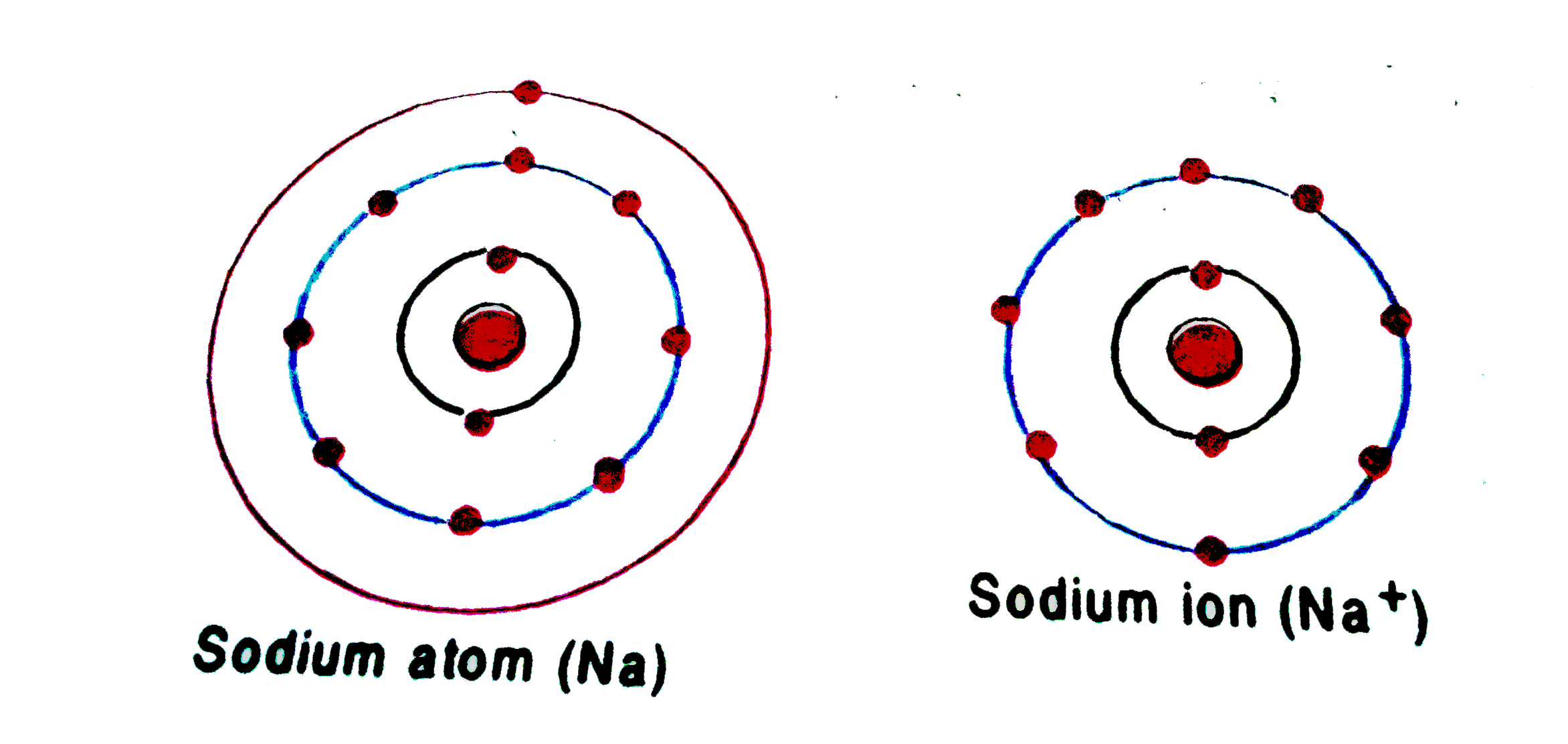
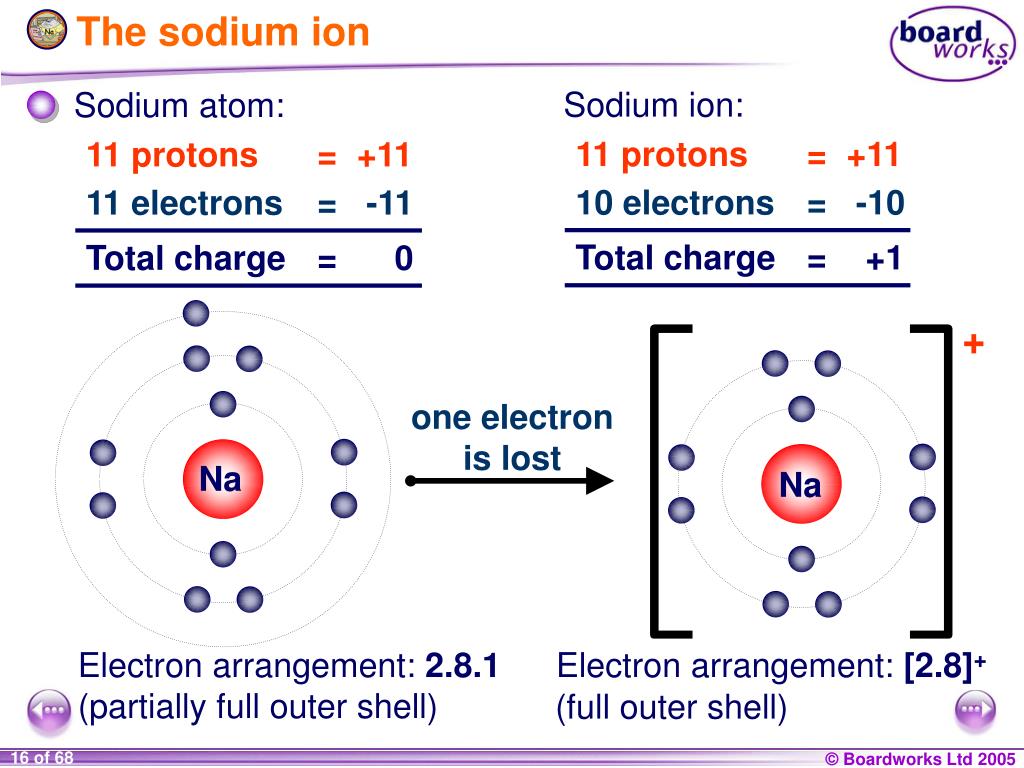
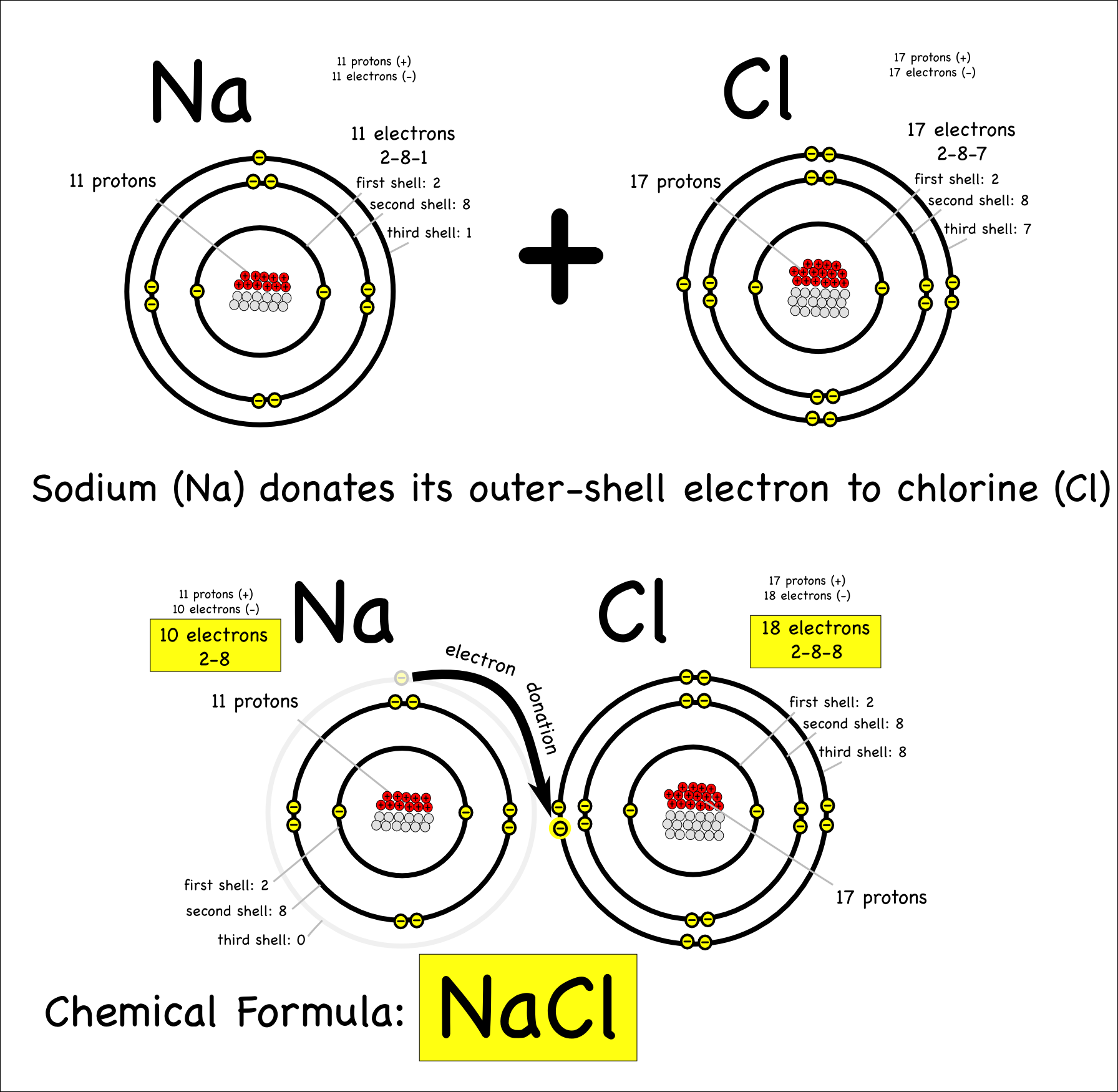
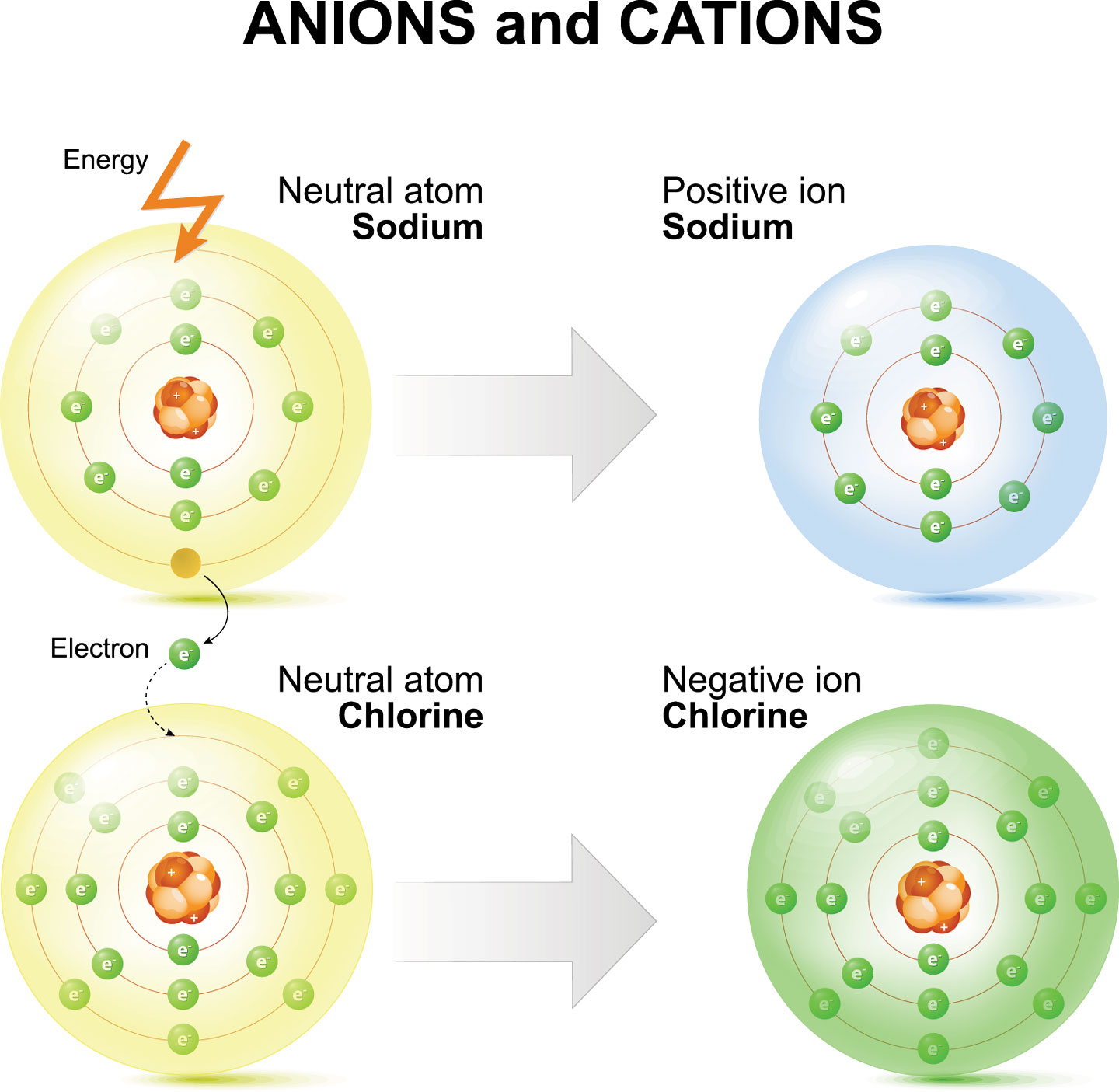
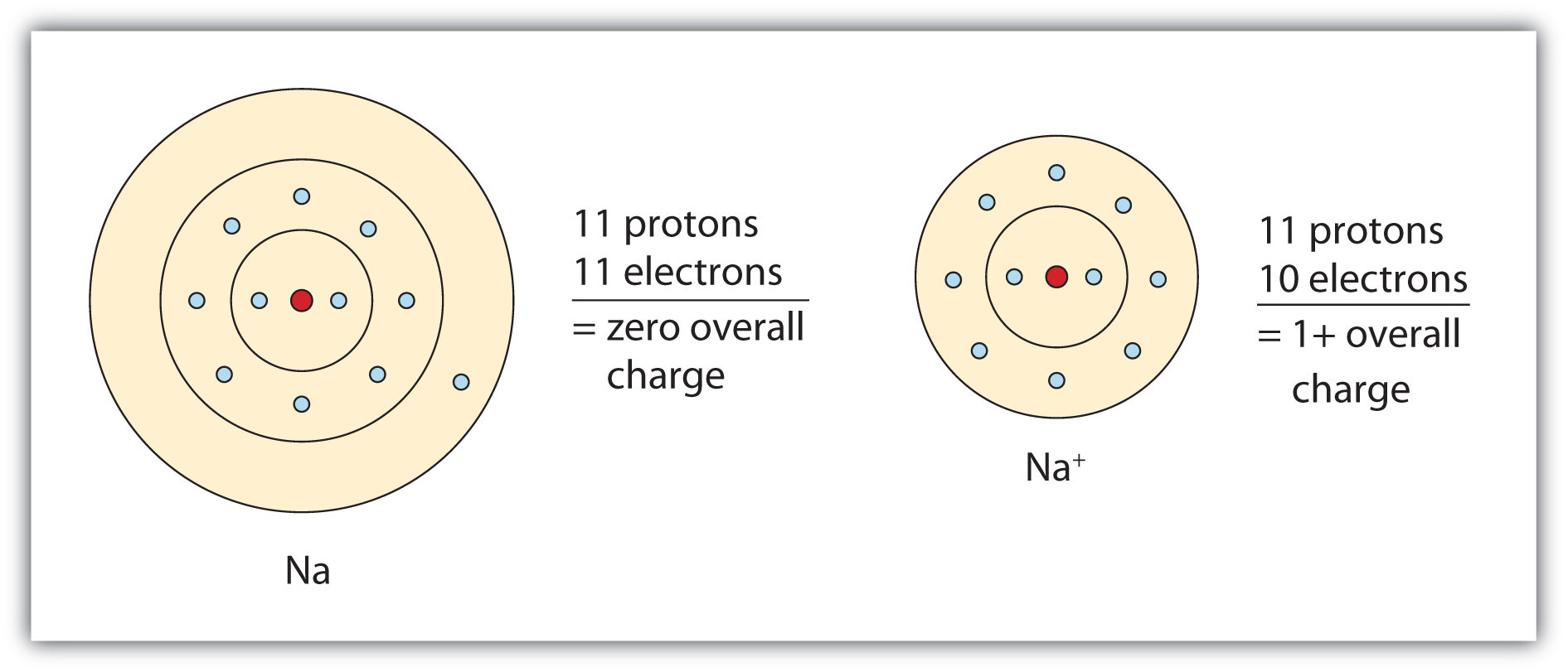
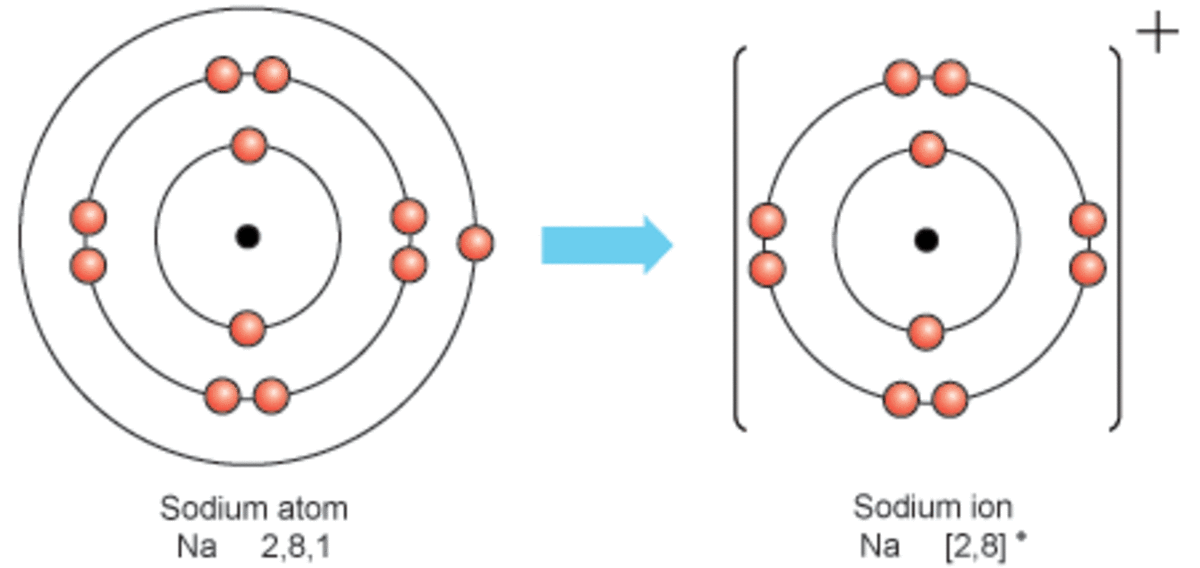
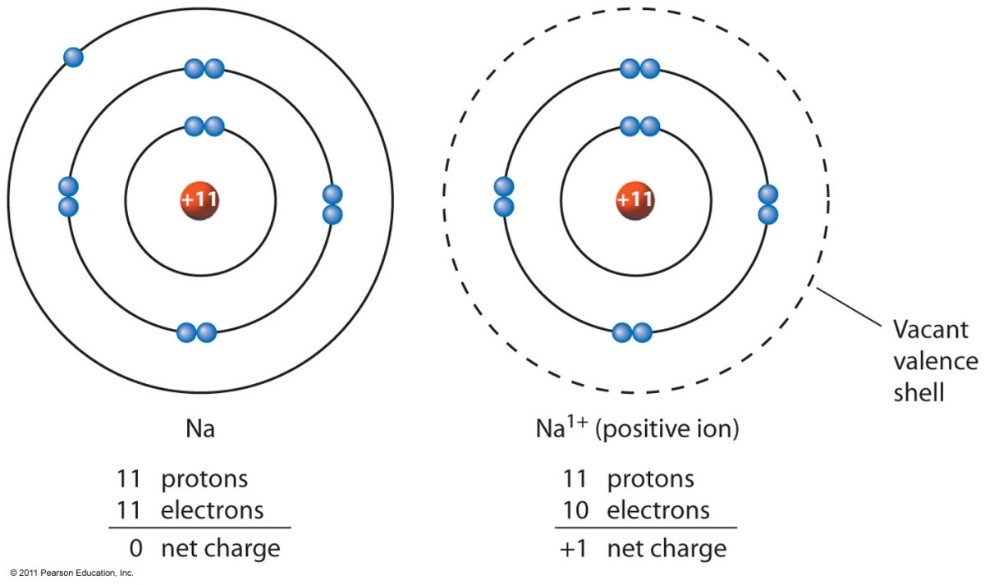
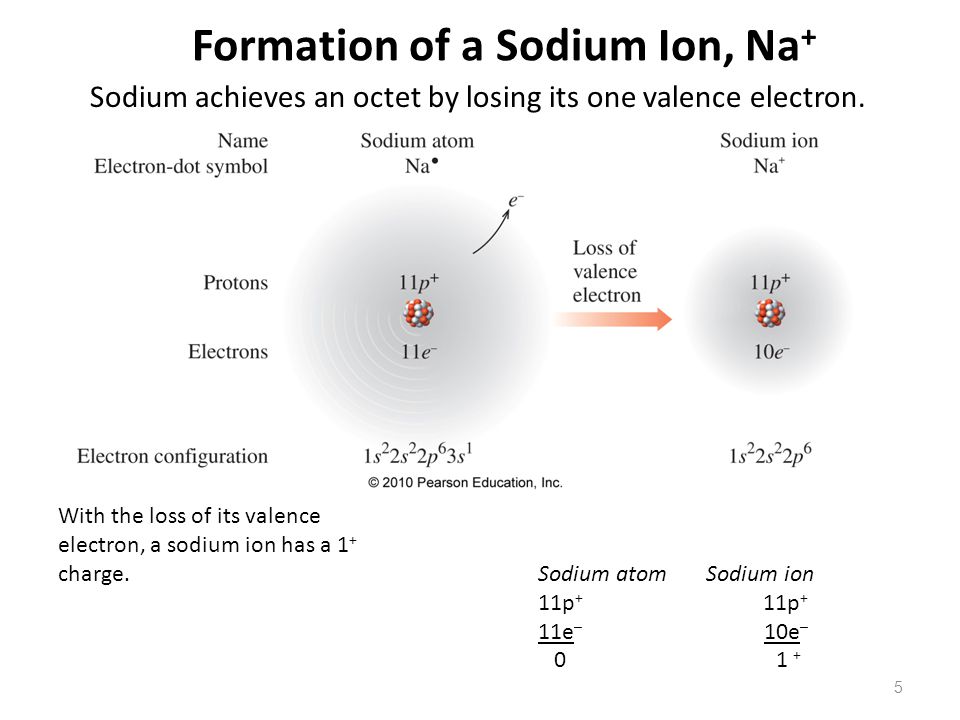
Sodium Forms An Ion With A Charge Of - The sodium atom loses its outer electron to become a sodium ion. Well, we form a na^+ ion. For example, in the compound sodium chloride — table salt — the sodium. Sodium metal is easily oxidized. The sodium ion still has. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge.
Sodium metal is easily oxidized. The sodium atom loses its outer electron to become a sodium ion. The sodium ion still has. For example, in the compound sodium chloride — table salt — the sodium. Well, we form a na^+ ion. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge.
The sodium atom loses its outer electron to become a sodium ion. Sodium metal is easily oxidized. Well, we form a na^+ ion. The sodium ion still has. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. For example, in the compound sodium chloride — table salt — the sodium.
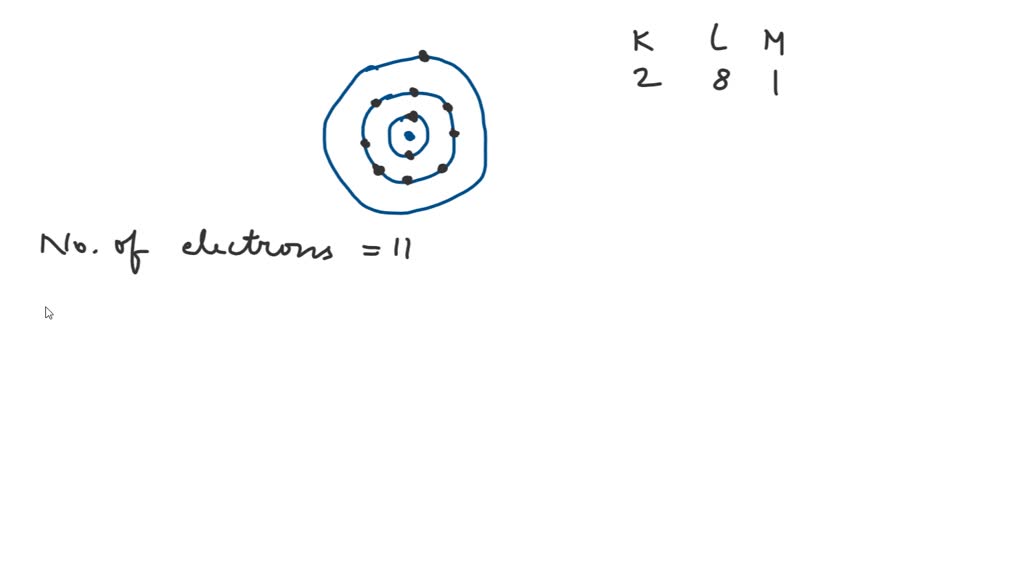
Sodium Electron Configuration Electron Configuration Sodium What is
The sodium atom loses its outer electron to become a sodium ion. The sodium ion still has. For example, in the compound sodium chloride — table salt — the sodium. Sodium metal is easily oxidized. Well, we form a na^+ ion.
PPT KS4 Chemistry PowerPoint Presentation, free download ID5413898
For example, in the compound sodium chloride — table salt — the sodium. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. Sodium metal is easily oxidized. The sodium atom loses its outer electron to become a sodium ion. The sodium ion still has.
Sodium Forms an Ion With a Charge of JasminehasGillespie
Well, we form a na^+ ion. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. Sodium metal is easily oxidized. For example, in the compound sodium chloride — table salt — the sodium. The sodium ion still has.
subatomic particles Montessori Muddle
Sodium metal is easily oxidized. The sodium ion still has. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. The sodium atom loses its outer electron to become a sodium ion. For example, in the compound sodium chloride — table salt — the sodium.
SOLVED Sodium has 11 electrons arranged in three energy levels. In
Well, we form a na^+ ion. The sodium atom loses its outer electron to become a sodium ion. The sodium ion still has. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. For example, in the compound sodium chloride — table salt — the sodium.
Explainer Ions and radicals in our world Science News for Students
For example, in the compound sodium chloride — table salt — the sodium. Well, we form a na^+ ion. The sodium ion still has. The sodium atom loses its outer electron to become a sodium ion. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge.
Ions
Sodium metal is easily oxidized. Well, we form a na^+ ion. The sodium atom loses its outer electron to become a sodium ion. The sodium ion still has. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge.
Chemical Bonding How Do Atoms Combine? What Are the Forces That Bind
The sodium ion still has. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. Sodium metal is easily oxidized. The sodium atom loses its outer electron to become a sodium ion. Well, we form a na^+ ion.
Ions Types, Summary, Classification & Facts
Well, we form a na^+ ion. The sodium ion still has. Sodium metal is easily oxidized. For example, in the compound sodium chloride — table salt — the sodium. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge.
Sodium Electron Configuration (Na) with Orbital Diagram
Well, we form a na^+ ion. The sodium ion still has. The sodium atom loses its outer electron to become a sodium ion. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. Sodium metal is easily oxidized.
Well, We Form A Na^+ Ion.
Sodium metal is easily oxidized. When sodium atoms form ions, they always form a 1+ charge, never a 2+ or 3+ or even 1− charge. For example, in the compound sodium chloride — table salt — the sodium. The sodium ion still has.